State Regulation of Prescription Drugs in the United States
States can influence how health care is delivered to their citizens
Photo: iStock.com/Moussa81
Individual states have often served as proving grounds for programs that seek to modify how health care is delivered and funded. State governments and state legislatures have many reasons to be deeply focused on the health delivery system. First, they have a close relationship with their constituents and citizens and seek to reflect the specific characteristics of health care in their state. Second, they are often one of the largest employers in the state, with generous and comprehensive coverage that is a large part of the state budget. Third, they are financially responsible for Medicaid, workers’ compensation, retiree costs, public health functions and the justice-involved population—often under sharply constrained budget conditions. And last, they oversee the financial status of the health insurers that provide health coverage to private purchasers.
Consequently, some states are actively pursuing changes to the way pharmacy benefit managers (PBMs) operate in their states and how insurers design and administer pharmacy benefits. At the same time, they also actively are exploring purchasing options in an effort to reduce costs, increase access and improve health outcomes. In 2018, there were 32 bills passed in 28 states that involved the regulation of PBMs; in 2019, 47 states introduced 275 bills aimed at controlling prescription drug pricing. Some states are considering carving out the pharmacy benefit entirely for managed Medicaid populations to be administered by the state. This could give the state government more direct influence over pricing, dispensing rules and rebate negotiations as well as the preferred drug list and utilization management programs.
State Legislation
There are several ways state governments affect changes to the regulation of prescription drugs. Often the method is by legislative action—a process with many steps that can benefit from actuarial input. As a bill is being drafted, actuarial input may be sought to clarify language, or there may be a fiscal impact statement prepared if the bill would result in a change to the state budget. During the hearing and amendment stage of legislation, actuaries may prepare testimony, estimates of financial impacts or offer insights on potential language changes. After legislation passes, actuaries may be included in any necessary rulemaking as the guiding language of the bill is transformed into steps and methods of administrative rules.
The challenge with tracking state actions is that there are 50 states with different legislative schedules and levels of engagement. UC Hastings College of Law created a public repository of health law and action information, including legislative and regulatory materials.1 It identified areas of pharmaceutical interest to include pricing, formulary, PBM regulation, pharmacy regulation, clawback restrictions, biosimilars, generic blocks and delays. Other sources include the National Conference of State Legislatures and the National Academy for State Health Policy.
Price Transparency
One area of active interest for state governments concerns price transparency, a topic for which some state stakeholders and voters express great passion. Some express deep anxiety about the nature and the extent of the price spread between what PBMs collect from payers for a drug and what pharmacies are reimbursed. Some state governments require that PBMs reveal much more about the actual cost of drugs, rebate arrangements, spreads in pricing and any financial arrangements between PBMs and manufacturers. They restrict the use of “gag rules,” so pharmacists may not be restricted from informing a beneficiary if there is an option to obtain a prescribed drug more cheaply than under their benefit plans. Additionally, some states require PBMs to post prices or to provide public announcements of price changes, either by data feeds to the state or by operating public websites.
Utilization Management
Some laws and rules address utilization management methods, such as step therapy, prior authorization or the use of narrow networks. Patients and doctors have concerns about losing access to some drug regimens, and some state governments have responded with new regulations that put restrictions on how and when formulary changes can be made as well as stipulating exceptions to any rules.
Member Cost-Share and Benefit Design
Other states are addressing member cost-share and benefit designs, which have become more important as drug costs escalate and members bear more of the cost. However, a state’s ability to manage employee benefits can often be limited due to the express preemption provision of the Employee Retirement Income Security Act of 1974 (ERISA). This provision preempts all state laws that relate to ERISA-governed employee benefit plans to prevent a patchwork of state regulations.
Other issues arise when the balance between affordability and control are not well understood, and adjusting components of the pharmacy delivery system can have unintended consequences. Some studies have shown that the more fractured the components of a delivery system are, the harder it is to deliver coordinated care to patients with multiple touchpoints in the delivery system.
Community Pharmacies
Another state constituency is composed of local business groups, towns and counties, particularly in underserved regions. As major PBMs have grown in strength and integrated with large drugstore chains and health plans, community pharmacies express dismay at the perceived erosion in their reimbursements and independence.
State legislative action is sometimes aimed at maintaining the financial well-being of smaller pharmacies through “any willing provider” legislation or minimum dispensing fees. An example of this is a 2015 Arkansas law that prohibits PBMs from reimbursing pharmacies at a lower rate than the cost to dispense the drug, and it allows pharmacies to refuse to dispense the drug if the maximum amount the plan will pay for the drug is too low.
Managing Unit Costs and Trends
States are major purchasers of drugs, and like other payers, they seek to manage unit costs and trends that stretch their abilities to raise funds to pay benefits. While in many cases the idea is to manage the costs for those expenses in the state budget, such as Medicaid or state employee benefit plans, states also seek to extend savings to residents and other constituents.
Many states can and do retain control of the lists of drugs covered for benefits delivered in programs where the state is the payer, such as state employee benefit plans or Medicaid programs. A statewide preferred drug list determines which drugs are covered and under which benefit conditions.
Some states have moved to increase the state government control. For instance, Kentucky (proposed) and West Virginia (enacted) carve out the drug benefit from the Medicaid managed care program and return it to a fee-for-service payment method, with the state more actively involved in contracting for manufacturer rebates and pharmacy discounts.
One of the more interesting areas of activity for states seeking to manage pharmaceutical costs is to begin direct contracting for all or some therapies. This is illustrated by recent developments in the cure of some forms of Hepatitis C. While prices have come down, the cost of treatments is still a significant burden due to the prevalence of the disease in the populations for which the states have responsibility, such as Medicaid and those involved with the justice system. Court rulings have made it clear that states cannot restrict payment for care. Louisiana and Washington have developed solutions by contracting directly with a pharmaceutical manufacturing partner.2,3
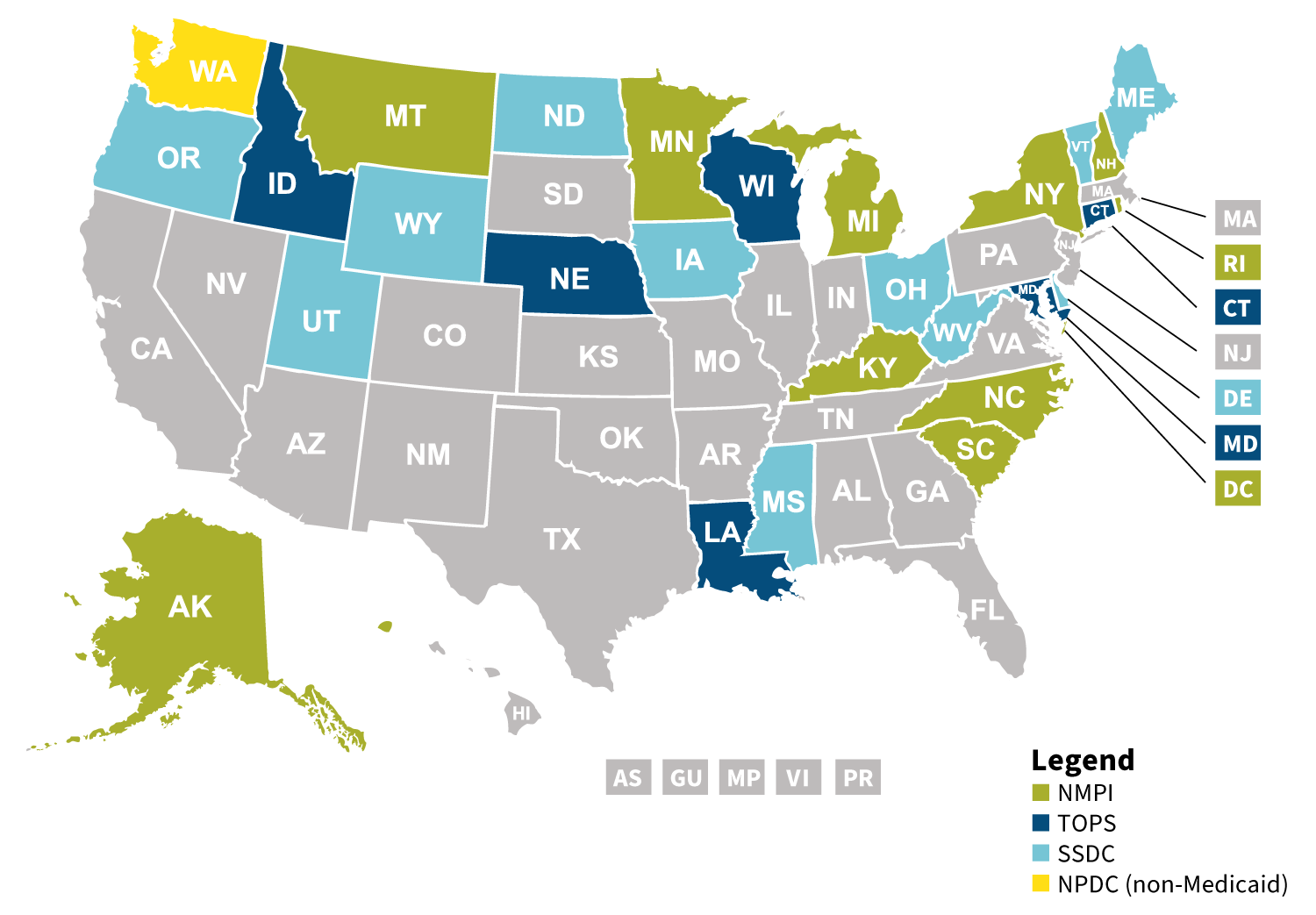
Bulk purchasing is another state method that seeks to control the costs and trends in pharmacy. As of 2019, there were five pools operating (see Figure 1).
Figure 1: Pharmaceutical Multistate Bulk Purchasing as of 2019
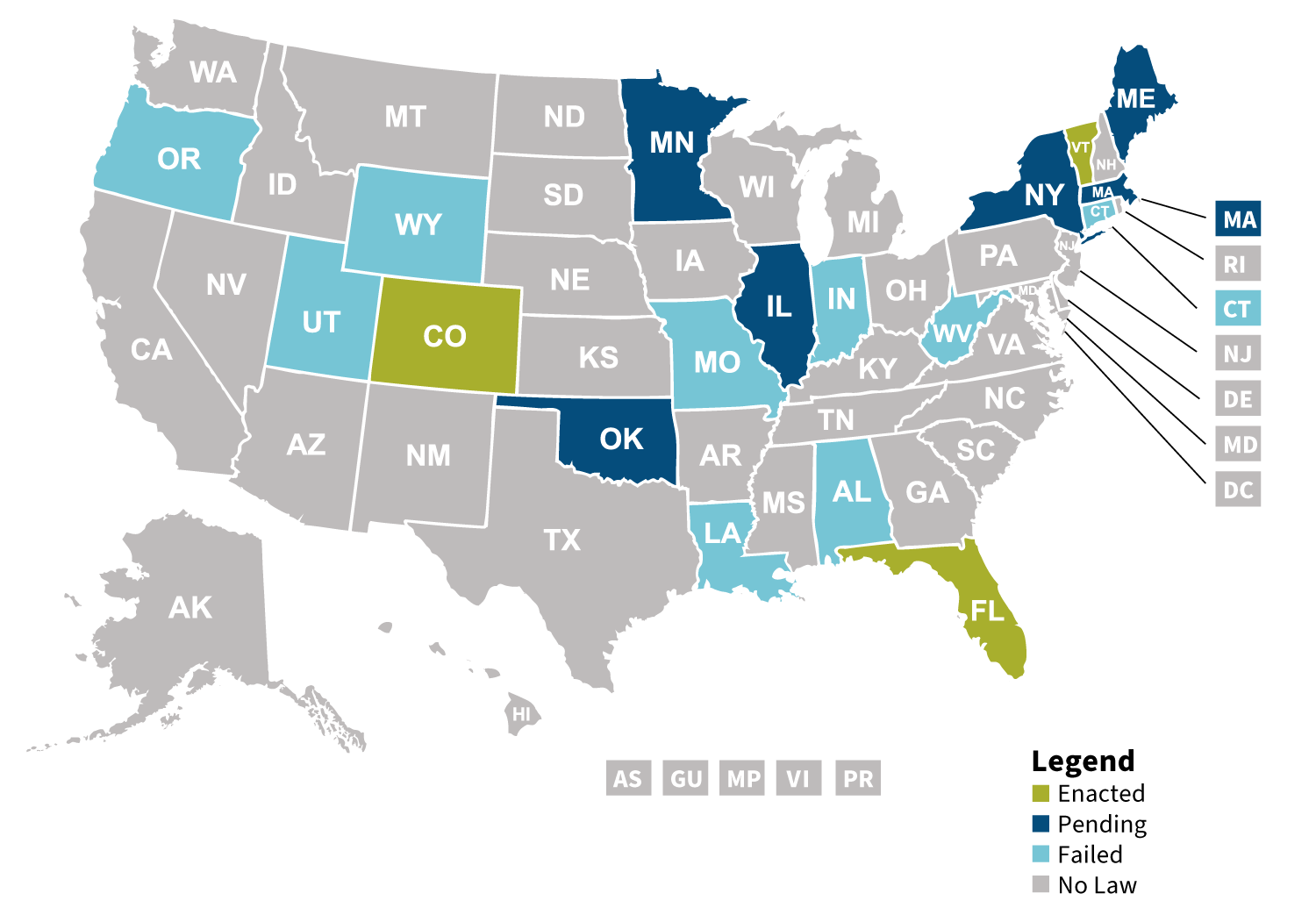
Some states are seeking to directly import drugs, something that may not be successful if advantageous contracts are put at risk for the country of origin (see Figure 2).
Figure 2: 2019 State Legislation for Prescription Drug Importation
California is considering taking state involvement in the management of pharmaceutical care costs to the ultimate point of manufacturing generics under the label CalRx.4 This was included in the 2020 state budget proposal and reflects the extent to which states are concerned with managing pharmacy costs.
States also are considering how to manage the financial well-being of small employer groups and individual markets as exceptionally high-cost therapies emerge. One of the concepts is to provide some form of risk-share or reinsurance pool at a statewide level.
However, state actions are not the final word. This fall, the Supreme Court heard arguments on an Arkansas state law regulating pharmacy reimbursement by PBMs. It is the first time the Supreme Court has ruled on a case involving PBMs, but as states become more active in controlling the pharmaceutical delivery system, it may not be the last. The outcome of the hearing will have implications on the ability of states to regulate PBMs.
Conclusion
Changing regulations in the pharmaceutical delivery system are happening at every level of government. Even though there are active efforts to make national changes in the pricing of pharmaceuticals, the states’ role in managing pharmacy benefit costs and delivery will continue to be active and innovative.
States have the ability to reflect the local delivery system, have a compelling budgetary need to control spending, and they often have greater flexibility than the federal government to pilot new programs. Some states are particularly innovative and operate as laboratories for health policy. These states are worth watching, particularly with respect to pharmaceuticals, because successes spread and failures can be useful learning tools.
Statements of fact and opinions expressed herein are those of the individual authors and are not necessarily those of the Society of Actuaries or the respective authors’ employers.
References:
- 1. The Source on Healthcare Price & Competition. The Database of State Laws Impacting Healthcare Cost and Quality. UC Hastings College of Law (accessed December 16, 2020). ↩
- 2. Phillips, Courtney N. Gilead’s Asegua Therapeutics Selected to Partner With Health Department for Hepatitis C Drug Subscription Model. Louisiana Department of Health, March 26, 2019 (accessed December 16, 2020). ↩
- 3. Washington State Health Care Authority. Health Care Authority Announces AbbVie US LLC as Apparently Successful Bidder for Hepatitis C Virus Treatment Drug. Washington State Health Care Authority, April 26, 2019 (accessed December 16, 2020). ↩
- 4. Office of Governor Gavin Newsom. Governor Newsom Proposes 2020-21 State Budget. State of California, January 10, 2020 (accessed December 15, 2020). ↩
Copyright © 2021 by the Society of Actuaries, Chicago, Illinois.

